Abstract
Background
Sex-determining region Y-box 3 (SOX3) protein, a SOX transcriptions factors group, has been identified as a key regulator in several diseases, including cancer. Downregulation of transcriptions factors in invasive ductal carcinoma (IDC) can interfere in neoplasia development, increasing its aggressiveness. We investigated SOX3 protein expression and its correlation with apoptosis in the MDA-MB-231 cell line, as SOX3 and Pro-Caspase-3 immunoexpression in paraffin-embedded invasive ductal carcinoma tissue samples from patients (n = 27). Breast cancer cell line MDA-MD-231 transfected with pEF1-SOX3 + and pEF1-Empty vector followed by cytotoxicity assay (MTT), Annexin-V FITC PI for apoptosis percentage assessment by flow cytometry, qPCR for apoptotic-related gene expression, immunofluorescence, and immunohistochemistry to SOX3 immunolocalization in culture cells, and paraffin-embedded invasive ductal carcinoma tissue samples.
Results
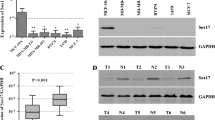
Apoptotic rate was higher in cells transfected with pEF1-SOX3 + (56%) than controls (10%). MDA-MB-231 transfected with pEF1-SOX3 + presented upregulation of pro-apoptotic mRNA from CASP3, CASP8, CASP9, and BAX genes, contrasting with downregulation antiapoptotic mRNA from BCL2, compared to non-transfected cells and cells transfected with pEF1-empty vector (p < 0.005). SOX3 protein nuclear expression was detected in 14% (4/27 cases) of ductal carcinoma cases, and pro-Caspase-3 expression was positive in 50% of the cases.
Conclusion
Data suggest that SOX3 transcription factor upregulates apoptosis in breast cancer cell line MDA-MB-231, and has a down nuclear expression in ductal carcinoma cases, and need to be investigated as a tumor suppressor protein, and its loss of expression and non-nuclear action turn the cells resistant to apoptosis. Further studies are necessary to understand how SOX3 protein regulates the promoter regions of genes involved in apoptosis.





Similar content being viewed by others
Data availability
Not applicable.
Abbreviations
- TFs:
-
Transcription factors
- IDC:
-
Invasive ductal carcinoma
- HMG:
-
High-mobility group
- EMT:
-
Epithelial mesenchymal transition
- IHC:
-
Immunohistochemistry
- DMEM:
-
Dulbecco’s modified eagle medium
- PBS:
-
Phosphate buffered saline
- GFP:
-
Green fluorescent protein
- BSA:
-
Bovine Serum Albumin
- MTT:
-
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide
- DMSO:
-
Dimethyl sulfoxide
- PI:
-
Propidium iodide
- UFMS:
-
Federal university of mato grosso do sul
References
Dai X, Xiang L, Li T, Bai Z. Cancer hallmarks, biomarkers and breast cancer molecular subtypes. J Cancer. 2016;7:1281–94.
El Hachem G, Gombos A, Awada A. Recent advances in understanding breast cancer and emerging therapies with a focus on luminal and triple-negative breast cancer. F1000Research. 2019;8.
Patten DK, Sharifi LK, Fazel M. New Approaches in the Management of Male Breast Cancer. Clin Breast Cancer. 2013;13:309–14.
Takahashi K, Yamanaka S. A decade of transcription factor-mediated reprogramming to pluripotency. Nat Rev Mol Cell Biol. 2016;17:183–93.
Gertz J, Reddy TE, Varley KE, Garabedian MJ, Myers RM. Genistein and bisphenol A exposure cause estrogen receptor 1 to bind thousands of sites in a cell type-specific manner. Genome Res. 2012;22:2153–62.
Sarkar A, Hochedlinger K. The sox family of transcription factors: versatile regulators of stem and progenitor cell fate. Cell Stem Cell. 2013;12:15–30.
Mehta GA, Khanna P, Gatza ML. Emerging role of SOX proteins in breast cancer development and maintenance. J Mammary Gland Biol Neoplasia. 2019;24:213–30.
Li K, Wang R-W, Jiang Y-G, Zou Y-B, Guo W. Overexpression of Sox3 is associated with diminished prognosis in esophageal squamous cell carcinoma. Ann Surg Oncol. 2013;20:459–66.
Yan Q, Wang F, Miao Y, Wu X. Sex-determining region Y-box3 (SOX3) functions as an oncogene in promoting epithelial ovarian cancer by targeting Src kinase. Tumor Biol. 2016;37:12263–71.
Guo Y, Yin J, Tang M, Yu X. Downregulation of SOX3 leads to the inhibition of the proliferation, migration and invasion of osteosarcoma cells. Int J Oncol. 2018;52:1277–84.
Cai QY, Liang GY, Zheng YF, Tan QY, Wang RW, Li K. Sox3 silencing inhibits metastasis and growth of esophageal squamous cell carcinoma cell via down-regulating GSK-3β. Int J Clin Exp Pathol. 2016;9:2939–49.
Acloque H, Ocaña OH, Matheu A, Rizzoti K, Wise C, Lovell-Badge R, et al. Reciprocal repression between Sox3 and Snail transcription factors defines embryonic territories at gastrulation. Dev Cell. 2011;21:546–58.
Comşa Ş, Cîmpean AM, Raica M. The story of MCF-7 breast cancer cell line: 40 years of experience in research. Anticancer Res. 2015;35:3147–54.
Holliday DL, Speirs V. Choosing the right cell line for breast cancer research. Breast Cancer Res. 2011;13:215.
Feng Y, Xiao F, Yang N, Zhu N, Fu Y, Zhang H, et al. Overexpression of Sox3 is associated with promoted tumor progression and poor prognosis in hepatocellular carcinoma. Int J Exp Pathol. 2017;10:7873–81.
Vicentic JM, Drakulic D, Garcia I, Vukovic V, Aldaz P. SOX3 can promote the malignant behavior of glioblastoma cells. Cell Oncol Cellular Oncol. 2018;42:41–54.
Song L, Liu D, He J, Wang X, Dai Z, Zhao Y, et al. SOX1 inhibits breast cancer cell growth and invasion through suppressing the Wnt/β-catenin signaling pathway. APMIS. 2016;124:547–55.
Das T, Nair RR, Green R, Padhee S, Howell M, Banerjee J, et al. Actinomycin D down-regulates SOX2 expression and induces death in breast cancer stem cells. Anticancer Res. 2017;37:1655–63.
Liang S, Furuhashi M, Nakane R, Nakazawa S, Goudarzi H, Ichi HJ, et al. Isolation and characterization of human breast cancer cells with SOX2 promoter activity. Biochem Biophys Res Commun. 2013;437:205–11.
Mehta GA, Parker JS, Silva GO, Hoadley KA, Perou CM, Gatza ML. Amplification of SOX4 promotes PI3K/Akt signaling in human breast cancer. Breast Cancer Res Treat. 2017;162:439–50.
Zhang J, Liang Q, Lei Y, Yao M, Li L, Gao X, et al. SOX4 induces epithelial-mesenchymal transition and contributes to breast cancer progression. Cancer Res. 2012;72:4597–608.
Oliemuller E, Kogata N, Bland P, Kriplani D, Daley F, Haider S, et al. SOX11 promotes invasive growth and ductal carcinoma in situ progression. J Pathol. 2017;243:193–207.
Oliemuller E, Newman R, Tsang SM, Foo S, Muirhead G, Noor F, et al. Sox11 promotes epithelial/mesenchymal hybrid state and alters tropism of invasive breast cancer cells. Elife. 2020;9:1–29.
Coutinho LM, Vieira EL, Dela Cruz C, Casalechi M, Teixeira AL, Del Puerto HL, et al. Apoptosis modulation by activin A and follistatin in human endometrial stromal cells. Gynecol Endocrinol. 2016;32:161–5.
Martin SJ, Reutelingsperger CPM, McGahon AJ, Rader JA, Van Schie RCAA, LaFace DM, et al. Early redistribution of plasma membrane phosphatidylserine is a general feature of apoptosis regardless of the initiating stimulus: Inhibition by overexpression of BCL-2 and Abl. J Exp Med. 1995;182:1545–56.
Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods. 2001;25:402–8.
Acknowledgements
We thank the patients and volunteers who contributed to this research. We thank Doctor Amy Milsted very much for reviewing the manuscript.
Funding
Research supported by Federal University of Minas Gerais (Edital PRPq05/20160); This study was part of a PhD thesis by F.H.H.S. at the Pathology Post-Graduation Program at Universidade Federal de Minas Gerais, PhD scholarship funded by CAPES.
Author information
Authors and Affiliations
Contributions
FHSS: data curation, formal analysis, investigation, writing—original draft. AU: resources, supervision, writing—review and editing. CPA: data curation, formal analysis, investigation, writing—review and editing. TSR: data curation, investigation, writing—review and editing. EMSF: resources, investigation. ASM: resources, investigation. ME: methodology. SG: methodology, resources. LOA: data curation, resources, methodology. LR: data curation, methodology. HWG: data curation, methodology. CAO: resources, methodology. RCR: patient’s recruitment and sample curation. ITB: data curation, investigation, writing—review and editing. GDC: resources, investigation. EF: data curation, formal analysis, resources, supervision. HLDP: Conceptualization, formal analysis, funding acquisition, methodology, project administration, resources, supervision, writing -review and editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors disclose no potential conflicts of interest.
Ethical approval and consent to participate
Informed consent was obtained from all the subjects before inclusion in the study, which was approved by the local Human Investigation Committee (CAAE 20636419.2.0000.0021).
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Silva, F.H.d., Underwood, A., Almeida, C.P. et al. Transcription factor SOX3 upregulated pro-apoptotic genes expression in human breast cancer. Med Oncol 39, 212 (2022). https://doi.org/10.1007/s12032-022-01758-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-022-01758-0